AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Violation aufbau principle8/15/2023 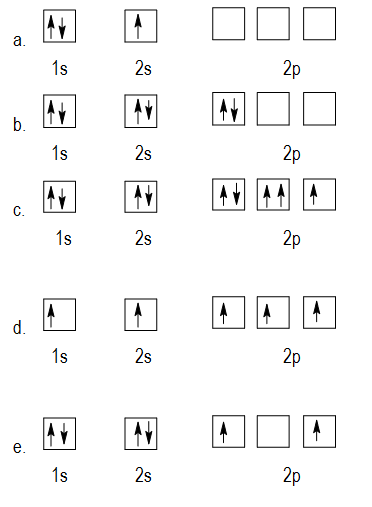
Otherwise they will have the same four quantum numbers, in violation of the Pauli Exclusion Principle. The distribution of electrons in the different orbitals can be done by following the various rules like the Hunds Rule, the Aufbaus Principle and the Paulis. According to Aufbau principle, electrons cannot be filled in higher orbitals, unless lower orbitals are completely occupied. Visually these two cases can be represented asĪs you can see, the 1 s and 2s subshells for beryllium atoms can hold only two electrons and when filled, the electrons must have opposite spins. If it is fully occupied, we have two \(m_s\) values, and the electron configuration is 1 s 2 (corresponding to helium). 
If the 1 s orbital contains only one electron, we have one \(m_s\) value and the electron configuration is written as 1 s 1 (corresponding to hydrogen). This tends to half filled d orbitals having greater stability and greater exchange energy. But here 1 electron migrate and 3d5 4s1 is seen experimentally. This is bcoz aufbau principle says that electrons should first b filled in orbitals hving lower energy so Cr should hav 3d4 4s2 configuration. Only two electrons can have these numbers, so that their spin moments must be either \(m_s = -1/2\) or \(m_s = 1/2\). 6 years ago Chromium violates aufbau principle. a) violates Hund's Rule, since electrons in 2p are doubled up before each of the orbitals at that energy have one in them b) violates the Aufbau principle, because the 1s orbital is missing an electron. 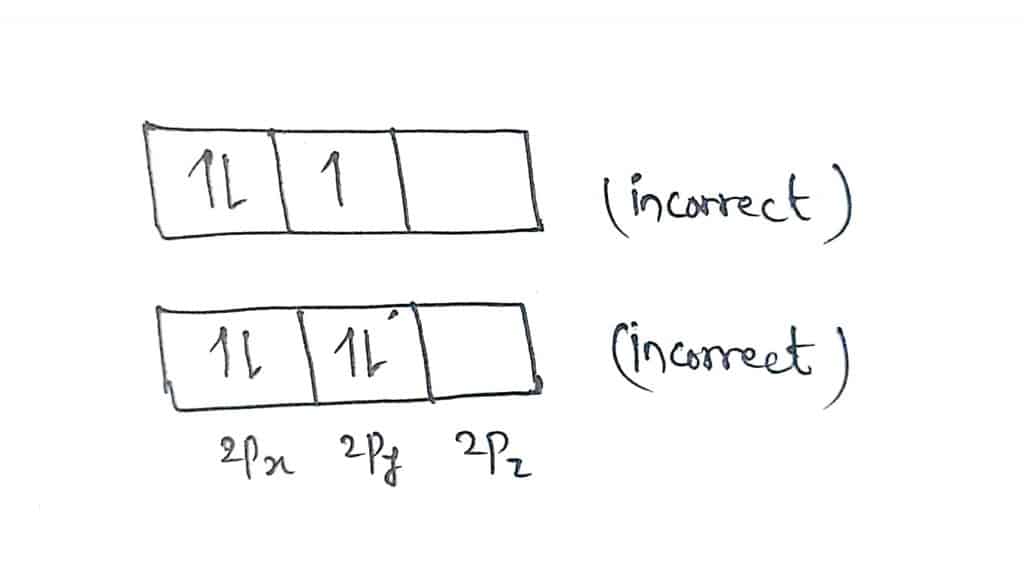
This means if one electron is assigned as a spin up ( 1/2) electron, the other electron must be spin-down (-1/2) electron.Įlectrons in the same orbital have the same first three quantum numbers, e.g., \(n=1\), \(l=0\), \(m_l=0\) for the 1 s subshell. The orbital diagram in which the Aufbau principle is violated is: The answer given for this question is 3, but according to me if asked for Hund's rule violation then it would be 1, as electrons are not distributed among the subshell in such a way to give maximum number of unpaired electron with parallel spin. As an orbital can contain a maximum of only two electrons, the two electrons must have opposing spins. The Pauli Exclusion Principle states that, in an atom or molecule, no two electrons can have the same four electronic quantum numbers. This electron configuration violates the Pauli exclusion principle, This electron configuration obeys Hunds rule Incorrect violates the Pauli exclusion.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
